Nerve-spring technique could achieve a functional trifecta outcome of robotic intracorporeal studer’s orthotopic neobladder in the male

Objectives: To summarize some key steps of functional improvement in robotic intracorporeal studer’s orthotopic neobladder (RISON) of males, especially for nerve-spring technique. We also presented the result of 1-year follow-up aimed to illustrate its functional trifecta outcomes.
Methods: Robotic radical cystectomy with intracorporeal studer’s orthotopic neobladder was performed on 33 male patients by the same surgeon from April 2018 to March 2019. Nerve-sparing technique had been used in 11 of the 33 patients. A prospectively maintained dataset was retrospectively searched and the related perioperative and follow-up data were analyzed. The functional trifecta outcomes referred to the freedom from recurrence, urinary continence and sexual function recovery after one year.
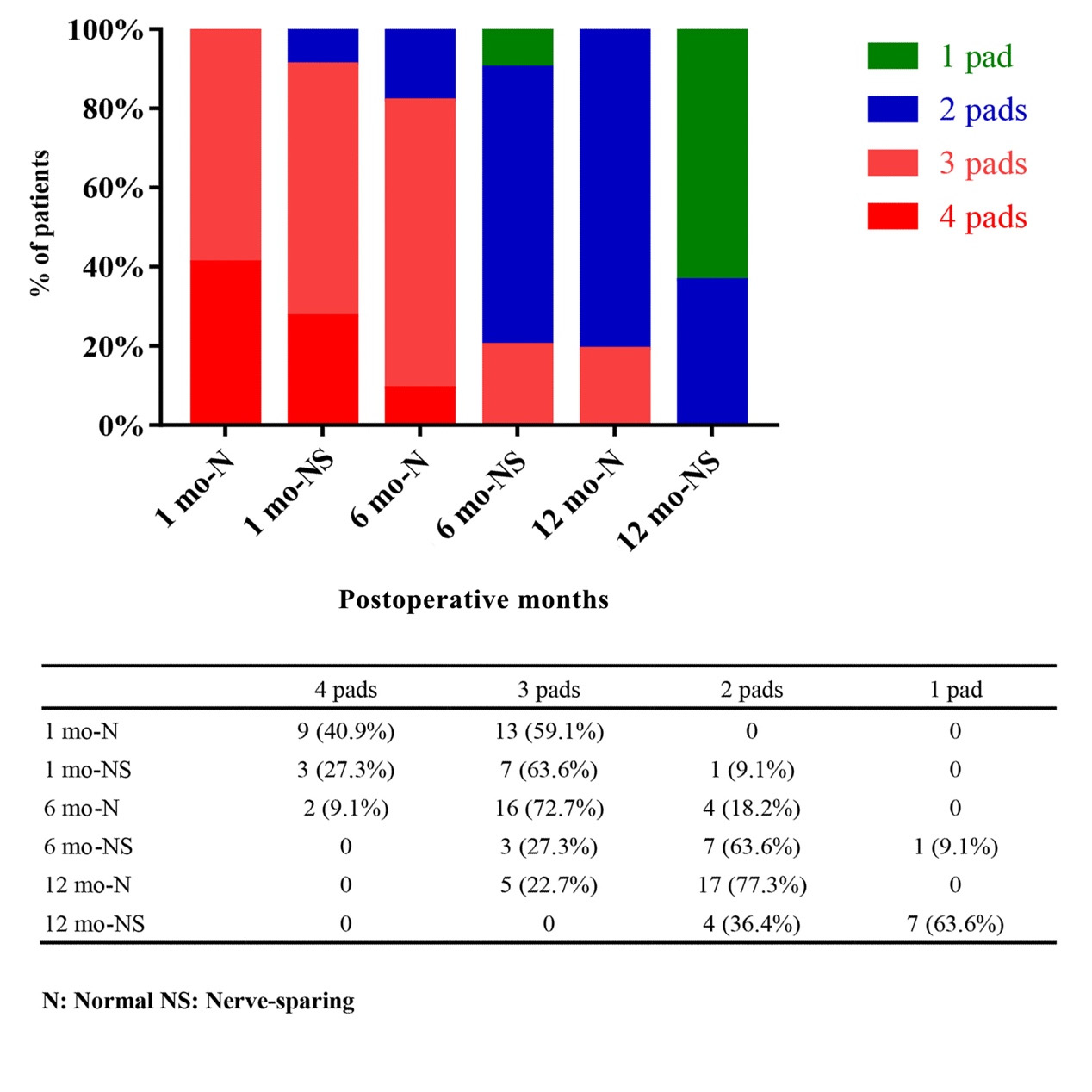
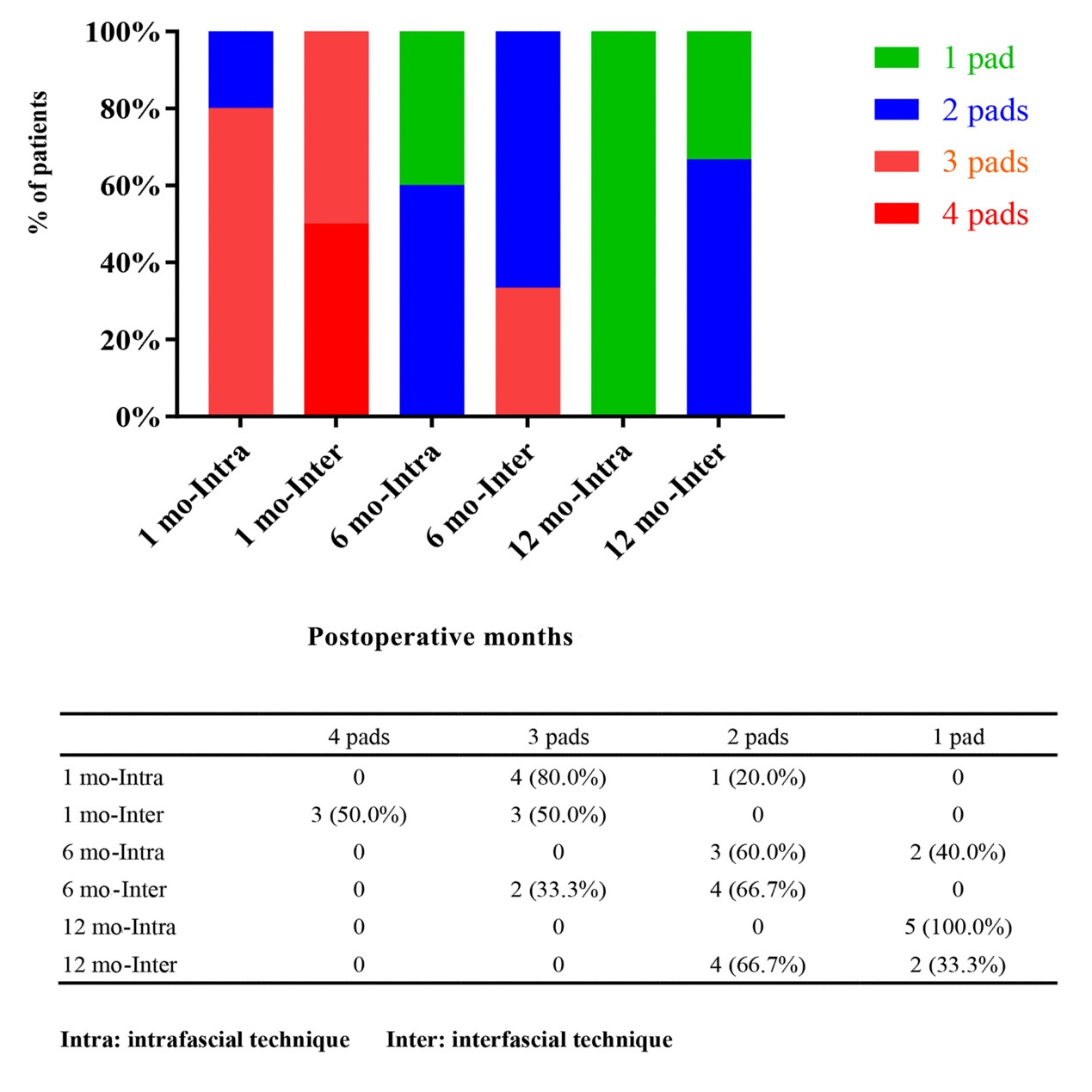
Results: A total of 33 males were included in our study. All perioperative information was recorded in detail. Thirty-two cases were confirmed to have negative surgical margin, except one pT3a case. And another case of incidental prostate cancer was diagnosed pathologically. All patients (100%) were recurrence-free one year after the operation. Eleven patients underwent nerve-sparing surgeries, including inter-fascial techniques or intra-fascial techniques. All these patients attained daytime continence (0 pad) at 1 month. With the nighttime continence, nerve-sparing group (2, 2 ,1) used fewer pads than other 22 cases (3, 3 ,2) at 1, 6 or 12 month(s) respectively. We defined urinary continence as 0 pad in daytime and no more than 1 pad in nighttime. The median preoperative score of International Index of Erectile Function (IIEF-6) in the 11 cases was 24. The sexual function recovery was defined as IIEF-6 > 20. The final trifecta rate was 54.5% and the median follow-up time lasted 17 months (range, 12 to 22 months).
Conclusions: RISON could be a safe and feasible choice of urinary diversion. Nerve sparing techniques might help the patients achieve a relatively higher functional trifecta rate.
Introduction
PATIENTS AND METHODS
Study Population
Indications
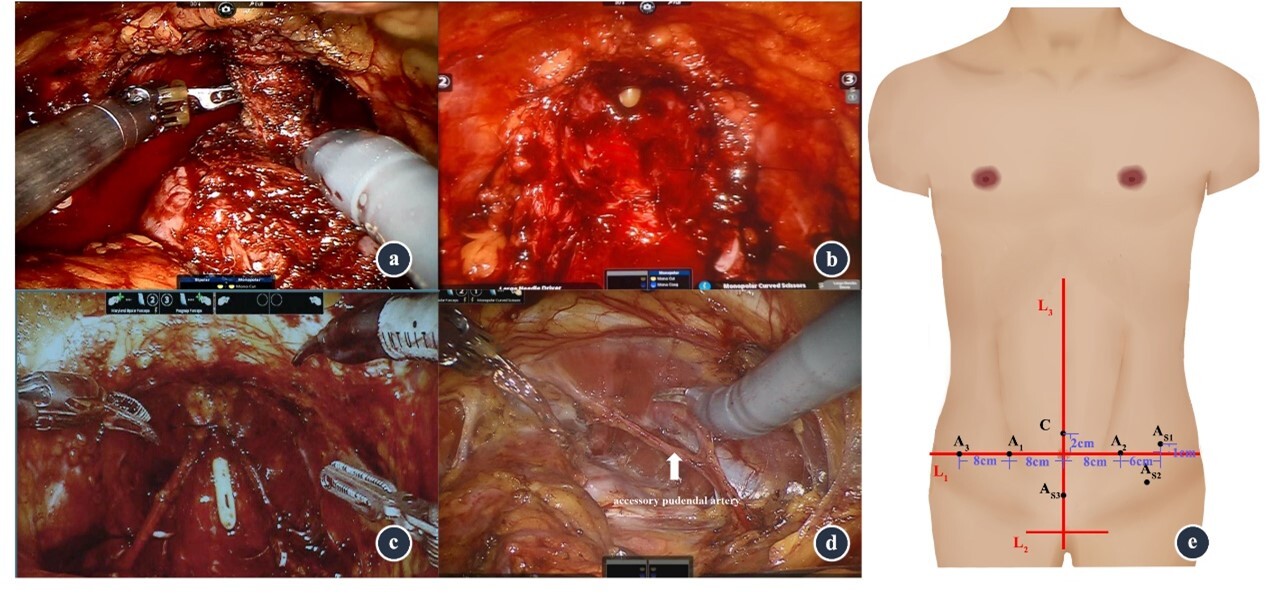
Perioperative care and surgical technique
Follow-up
Trifecta definition
Statistical Analysis
Results
DISCUSSION
| Complications | No nerve-sparing | Nerve-sparing | Treatment | P |
|---|---|---|---|---|
| Early (< 30 days, n and grade) | ||||
| Gastrointestinal complications | 4, I | 1, I | Oral medications or observation | |
| Infectious complications | 1, II | 1, II | Intravenous antibiotics | |
| Bleeding requiring transfusion | 1, II | 2, II | Transfusion | |
| Total | 6/22 (27.3%) | 4/11 (36.4%) | 1 | 1.000 |
| 10/33 (30.3%) | ||||
| Late (> 30 days, n and grade) | ||||
| Gastrointestinal complications | 3, I | 1, I | Oral medications or observation | |
| Infectious complications | 4, I | 2, I | Oral antibiotics | |
| Infectious complications | 3, II | 2, II | Intravenous antibiotics | |
| Pelvic cavity effusion | 1, III | 0 | Interventional ultrasound | |
| Total | 11/22 (50.0%) | 5/11 (45.5%) | 1.000 | |
| 16/33 (48.5%) | ||||
| Characteristics | No nerve-sparing | Nerve-sparing | P |
|---|---|---|---|
| Total, n | 22 | 11 | |
| Age (year), Median (IQR) | 58 (50‒63) | 53 (49‒57) | 0.089 |
| BMI, Kg/m2, Median (IQR) | 25.5 (20.3‒33.8) | 24.8 (22.9‒33.7) | < 0.001 |
| Charlson Comorbidity Index, n | 0.081 | ||
| 0‒2 | 5 (22.7%) | 1 (9.1 %) | |
| 3‒5 | 17 (77.3%) | 9 (81.8%) | |
| 6‒8 | 0 | 1 (9.1%) | |
| >8 | 0 | 0 | |
| Neoadjuvant Chemotherapy, n | 1.000 | ||
| Presence | 2 (9.1%) | 2 (18.2%) | |
| Absence | 20 (90.9%) | 9 (81.8%) | |
| Preoperative Continence | Normal | Normal | |
| Median Preoperative IIEF-6 score (IQR) | 19 (18‒20.2) | 24 (24‒25) | < 0.001 |
| Indwelling Gastric Tube, Days, Median (IQR) | 3 (3‒4) | 3 (2‒3) | < 0.001 |
| Tolerance Time, Days, Median (IQR) | 4.5 (3.75‒5.25) | 4 (3‒6) | 0.029 |
| Operation Time, Hours, Median (IQR) | 7 (5.38‒8.5) | 5 (4.5‒7) | < 0.001 |
| Blood Loss, ml, Median (IQR) | 250 (200‒500) | 200 (150‒300) | < 0.001 |
| Postoperative Hospital Stay, Days, Median (IQR) | 9 (8‒10.25) | 7 (7‒12) | < 0.001 |
|
Groups |
Nerve-sparing |
Intra-fascial |
Inter-fascial |
|---|---|---|---|
| Functional trifecta rate | 54.5% | 100% | 16.7% |
CONCLUSION
1. Messing EM, Catalona W. Urothelial tumors of the urinary tract. In: Campbell MF, Retik AB, Vaughan ED, Walsh PC, editors. Campbell’s urology. 7th ed. Philadelphia (PA): W. B. Saunders; 1998. pp. 2327–408.
2. Furrer MA, Studer UE, Gross T, Burkhard FC, Thalmann GN, Nguyen DP. Nerve-sparing radical cystectomy has a beneficial impact on urinary continence after orthotopic bladder substitution, which becomes even more apparent over time. BJU Int. 2018 Jun;121(6):935–44. https://doi.org/10.1111/bju.14123 PMID:29319917
3. Kessler TM, Burkhard FC, Perimenis P, Danuser H, Thalmann GN, Hochreiter WW, et al. Attempted nerve sparing surgery and age have a significant effect on urinary continence and erectile function after radical cystoprostatectomy and ileal orthotopic bladder substitution. J Urol. 2004 Oct;172(4 Pt 1):1323–7. https://doi.org/10.1097/01.ju.0000138249.31644.ec PMID:15371833
4. Bhatta Dhar N, Kessler TM, Mills RD, Burkhard F, Studer UE. Nerve-sparing radical cystectomy and orthotopic bladder replacement in female patients. Eur Urol. 2007 Oct;52(4):1006–14. https://doi.org/10.1016/j.eururo.2007.02.048 PMID:17360106
5. Aziz A, Gierth M, Rink M, Schmid M, Chun FK, Dahlem R, et al.; PROMETRICS 2011 Research Group. Optimizing outcome reporting after radical cystectomy for organ-confined urothelial carcinoma of the bladder using oncological trifecta and pentafecta. World J Urol. 2015 Dec;33(12):1945–50. https://doi.org/10.1007/s00345-015-1572-x PMID:25947885
6. Cacciamani GE, Winter M, Medina LG, Ashrafi AN, Miranda G, Tafuri A, et al. Radical cystectomy pentafecta: a proposal for standardisation of outcomes reporting following robot-assisted radical cystectomy. BJU Int. 2020 Jan;125(1):64–72. https://doi.org/10.1111/bju.14861 PMID:31260600
7. Asimakopoulos AD, Campagna A, Gakis G, Corona Montes VE, Piechaud T, Hoepffner JL, et al. Nerve Sparing, Robot-Assisted Radical Cystectomy with Intracorporeal Bladder Substitution in the Male. J Urol. 2016 Nov;196(5):1549–57. https://doi.org/10.1016/j.juro.2016.04.114 PMID:27423759
8. Spiess PE, Agarwal N, Bangs R, Boorjian SA, Buyyounouski MK, Clark PE, et al. Bladder Cancer, Version 5.2017, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2017 Oct;15(10):1240–67. https://doi.org/10.6004/jnccn.2017.0156 PMID:28982750
9. Alfred Witjes J, Lebret T, Compérat EM, Cowan NC, De Santis M, Bruins HM, et al. Updated 2016 EAU Guidelines on Muscle-invasive and Metastatic Bladder Cancer. Eur Urol. 2017 Mar;71(3):462–75. https://doi.org/10.1016/j.eururo.2016.06.020 PMID:27375033
10. Collins JW, Patel H, Adding C, Annerstedt M, Dasgupta P, Khan SM, et al. Enhanced Recovery After Robot-assisted Radical Cystectomy: EAU Robotic Urology Section Scientific Working Group Consensus View. Eur Urol. 2016 Oct;70(4):649–60. https://doi.org/10.1016/j.eururo.2016.05.020 PMID:27234997
11. Tyritzis SI, Hosseini A, Collins J, Nyberg T, Jonsson MN, Laurin O, et al. Oncologic, functional, and complications outcomes of robot-assisted radical cystectomy with totally intracorporeal neobladder diversion. Eur Urol. 2013 Nov;64(5):734–41. https://doi.org/10.1016/j.eururo.2013.05.050 PMID:23768634
12. Hernández V, Espinos EL, Dunn J, MacLennan S, Lam T, Yuan Y, et al. Oncological and functional outcomes of sexual function-preserving cystectomy compared with standard radical cystectomy in men: A systematic review. Urol Oncol. 2017 Sep;35(9):539.e17–29. https://doi.org/10.1016/j.urolonc.2017.04.013 PMID:28495555
13. Smith ZL, Christodouleas JP, Keefe SM, Malkowicz SB, Guzzo TJ. Bladder preservation in the treatment of muscle-invasive bladder cancer (MIBC): a review of the literature and a practical approach to therapy. BJU Int. 2013 Jul;112(1):13–25. https://doi.org/10.1111/j.1464-410X.2012.11762.x PMID:23356411
14. Kim JK, Park SY, Ahn HJ, Kim CS, Cho KS. Bladder cancer: analysis of multi-detector row helical CT enhancement pattern and accuracy in tumor detection and perivesical staging. Radiology. 2004 Jun;231(3):725–31. https://doi.org/10.1148/radiol.2313021253 PMID:15118111
15. Barentsz JO, Jager GJ, Witjes JA, Ruijs JH. Primary staging of urinary bladder carcinoma: the role of MRI and a comparison with CT. Eur Radiol. 1996;6(2):129–33. https://doi.org/10.1007/BF00181125 PMID:8797968
16. Ha HK, Koo PJ, Kim SJ. Diagnostic Accuracy of F-18 FDG PET/CT for Preoperative Lymph Node Staging in Newly Diagnosed Bladder Cancer Patients: A Systematic Review and Meta-Analysis. Oncology. 2018;95(1):31–8. https://doi.org/10.1159/000488200 PMID:29847834
17. Labbate C, Werntz RP, Adamic B, Steinberg GD. The Impact of Omission of Intraoperative Frozen Section Prior to Orthotopic Neobladder Reconstruction. J Urol. 2019 Oct;202(4):763–9. https://doi.org/10.1097/JU.0000000000000317 PMID:31059666
18. Lee H, Kim K, Hwang SI, Lee HJ, Byun SS, Lee SE, et al. Impact of prostatic apical shape and protrusion on early recovery of continence after robot-assisted radical prostatectomy. Urology. 2014 Oct;84(4):844–9. https://doi.org/10.1016/j.urology.2014.06.011 PMID:25129539
19. Paparel P, Akin O, Sandhu JS, Otero JR, Serio AM, Scardino PT, et al. Recovery of urinary continence after radical prostatectomy: association with urethral length and urethral fibrosis measured by preoperative and postoperative endorectal magnetic resonance imaging. Eur Urol. 2009 Mar;55(3):629–37. https://doi.org/10.1016/j.eururo.2008.08.057 PMID:18801612
20. el-Bahnasawy MS, Gomha MA, Shaaban AA. Urethral pressure profile following orthotopic neobladder: differences between nerve sparing and standard radical cystectomy techniques. J Urol. 2006 May;175(5):1759–63. https://doi.org/10.1016/S0022-5347(05)01019-0 PMID:16600753
21. Mungovan SF, Sandhu JS, Akin O, Smart NA, Graham PL, Patel MI. Preoperative Membranous Urethral Length Measurement and Continence Recovery Following Radical Prostatectomy: A Systematic Review and Meta-analysis. Eur Urol. 2017 Mar;71(3):368–78. https://doi.org/10.1016/j.eururo.2016.06.023 PMID:27394644
22. Nyame YA, Zargar H, Ramirez D, Ganesan V, Babbar P, Villers A, et al. Robotic-assisted Laparoscopic Bilateral Nerve Sparing and Apex Preserving Cystoprostatectomy in Young Men With Bladder Cancer. Urology. 2016 Aug;94:259–64. https://doi.org/10.1016/j.urology.2016.04.026 PMID:27132504
23. Walz J, Epstein JI, Ganzer R, Graefen M, Guazzoni G, Kaouk J, et al. A Critical Analysis of the Current Knowledge of Surgical Anatomy of the Prostate Related to Optimisation of Cancer Control and Preservation of Continence and Erection in Candidates for Radical Prostatectomy: an Update. Eur Urol. 2016 Aug;70(2):301–11. https://doi.org/10.1016/j.eururo.2016.01.026 PMID:26850969
24. Ganzer R, Stolzenburg JU, Neuhaus J, Weber F, Burger M, Bründl J. Is the striated urethral sphincter at risk by standard suture ligation of the dorsal vascular complex in radical prostatectomy? An anatomic study. Urology. 2014 Dec;84(6):1453–8. https://doi.org/10.1016/j.urology.2014.06.092 PMID:25432837
25. Wang X, Wu Y, Guo J, Chen H, Weng X, Liu X. Intrafascial nerve-sparing radical prostatectomy improves patients’ postoperative continence recovery and erectile function: A pooled analysis based on available literatures. Medicine (Baltimore). 2018 Jul;97(29):e11297. https://doi.org/10.1097/MD.0000000000011297 PMID:30024505
26. Weng H, Zeng XT, Li S, Meng XY, Shi MJ, He DL, et al. Intrafascial versus interfascial nerve sparing in radical prostatectomy for localized prostate cancer: a systematic review and meta-analysis. Sci Rep. 2017 Sep;7(1):11454. https://doi.org/10.1038/s41598-017-11878-7 PMID:28904361
27. Walz J, Burnett AL, Costello AJ, Eastham JA, Graefen M, Guillonneau B, et al. A critical analysis of the current knowledge of surgical anatomy related to optimization of cancer control and preservation of continence and erection in candidates for radical prostatectomy. Eur Urol. 2010 Feb;57(2):179–92. https://doi.org/10.1016/j.eururo.2009.11.009 PMID:19931974
28. Williams SB, Morales BE, Huynh LM, Osann K, Skarecky DW, Ahlering TE. Analysis of Accessory Pudendal Artery Transection on Erections During Robot-Assisted Radical Prostatectomy. J Endourol. 2017 Nov;31(11):1170–5. https://doi.org/10.1089/end.2017.0542 PMID:28859491
29. Henry BM, Pękala PA, Vikse J, Sanna B, Skinningsrud B, Saganiak K,et al. Variations in the Arterial Blood Supply to the Penis and the Accessory Pudendal Artery: A Meta-Analysis and Review of Implications in Radical Prostatectomy. J Urol. 2017; 198(2):345-353. https://doi.org/10.1016/j.juro.2017.01.080. PMID: 28202357

