Introduction
Stress urinary incontinence (SUI) refers to the involuntary urine leakage from the external urethral orifice when abdominal pressure is increased, during sneezing or coughing etc.. Pathophysiologically, in most patients with mild or moderate SUI, their urethral intrinsic sphincter is functionally impaired [
1]. In woman, delivery and surgery are the main causes of impaired function of urinary tract intrinsic sphincter, pelvic floor muscles and connective tissues, and the symptoms deteriorate with aging and accompanied hormonal changes [
2]. As with normal skeletal muscles, proper training can increase the strength and size of the sphincter. Kegel exercise is exactly based on this concept. Most SUI can be effectively treated by enhancing pelvic floor muscles and external urethral sphincter function [
3]. Nonetheless, since training course is long, some patients fail to adhere to the training schedule or plan, or some are unable to produce independent muscle contraction under some pathological conditions, as a result, failing to achieve the desired effect of Kegel training. Physiologically, both active muscle contraction and electrically-induced (passive) muscle contraction can equally achieve the training effect. Since the pelvic floor muscle fibers are located in the deep layer of the epidermis or mucosa, the traditional transcutaneous, vaginal and rectal electrical stimulation work only on the receptors of the surface layer or on mucosa and may cause discomfort. For this reason, we developed a miniature implantable wireless nerve stimulator (NuStim
®) that can be accurately implanted into the pelvic floor target muscles through minimally invasive surgery. By direct electrical stimulation, strong muscle contraction can be induced without autonomous contractions. The power and controlled signals of the stimulator are delivered by wireless transmittor free of any percutaneous electrode wires for connection, thereby avoiding any risk of shedding or infection associated with the wire connection.
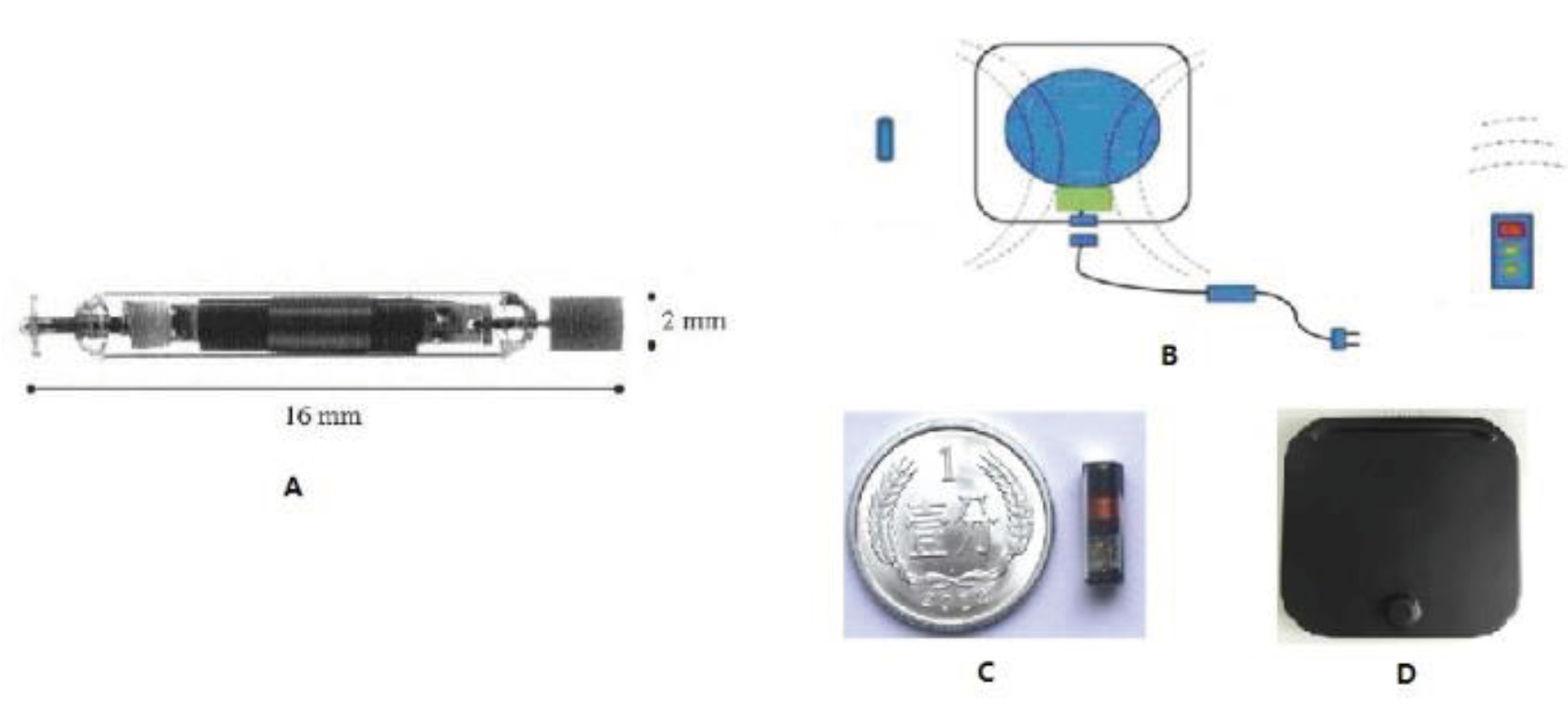
Muscle autonomic contraction and passive electrical stimulation have been extensively and intensively studied, including use of an implantable miniature neurostimulator (BION,
Fig. 1A), similar to our product. The BION stimulator has been CE-certified in Europe and it treats acute urinary incontinence by stimulating the pudendal nerve [
4]. However, compared to the NuStim neurostimulator, BION is battery-powered and larger-sized. On the other hand, NuStim is cylindrical and is only 3 mm in diameter and 10 mm in length.
To verify the safety and effectiveness of NuStim, we conducted an animal study with NuStim. In the study, the NuStim stimulator was implanted into the beagle dog to confirm its biosafety and the tissue compatibility with the tissues of the recipient animals, and to verify the stability and the effectiveness of the system.
Materials and methods
Design of the device
The NuStim device (General Stim Inc., Zhejiang, China) consisted of the following three components: a stimulator (Fig. 1C), an Android pad (APP) and a radio-frequency (RF) cushion (Fig. 1D). Stimulation parameters were set through the APP and sent to the RF cushion (Fig. 1B), which coupled energy to the NuStim wireless neurostimulator with a pulse amplitude of 20 grades, 0~16 V (adjustable), frequency of 4 ‒ 50 Hz, pulse width of 40–450 μs, depending on the magnitude level.
Experimental procedures
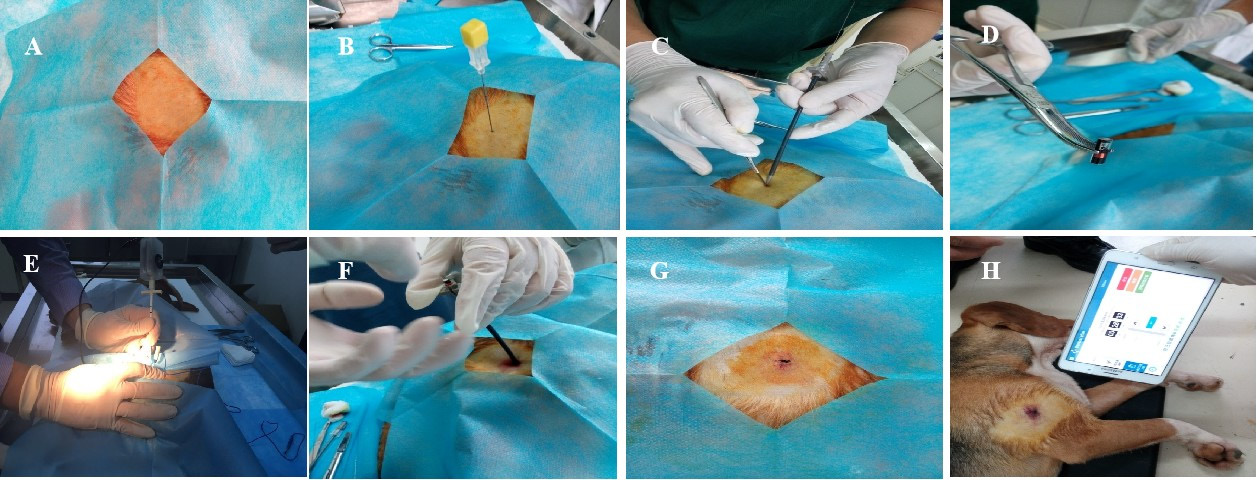
Three healthy adult male beagles (weighing 9‒11 kg, aged 6 months) were used in the experiment. This experiment was approved by the Animal Experimental Ethics Committee of Capital Medical University (No. AEEI-2015-115), Beijing, China. The implantation was performed in China Rehabilitation Center. The essential operation steps were as follows: (1) The animals were fasted 12 hours before surgery and weighed before anesthesia; (2) 2.5% pentobarbita was transabdominally administered, depending on the animal body weight (at 25 mg/kg); (3) The stimulator was implanted into the quadriceps femoris of each dog. In brief, the implantation was carried out as follows (Fig. 2): The animals were placed in the lateral position, disinfected and properly drapped after shaving. The quadriceps were located by pressing the thigh bone, and then pushed vertically into the skin by using a puncture needle. The test stimulator was set at 3 V for temporary use in vitro, and the needle was pushed forward until the leg muscle contraction was visible. The needle depth was fine-tuned to reach a point where minimal stimulation could produce the maximal contraction (so-called “threshold stimulus target location”). The (test stimulator) dilator tube and the puncture needle were then pulled out. After the NuStim implantable stimulator was put in, it was pushed by means of the puncture needle of the NuStim-specific surgical tool. The target position was re-confirmed by adjusting the test stimulator and observing the contraction. The test stimulator propulsion needle was kept still while the sheath was slowly removed until the NuStim implant was completely implanted into the body; (4) The incision wound was closed and the animals were put back to the cage; (5) The wound was disinfected regularly and antibiotic was given intramuscularly to ensure good recovery.
Figure 1. Schematic diagram of BION and NuStim devices
Figure 2. Operation steps: A. Shaving and disinfection at the surgical site; B. Puncture into the quadriceps femoris; C. Minimally invasive incision and skin expansion; D. A stimulator ready for implantation; E. Inoperative thresholding test; F. Stimulator implantation; G. Sutured wound; H. Postoperative functional assay.
Start-on test and daily training
First, the test was conducted one week after the operation. The animals were made to lie on the RF cushion and hypnotic was given. The stimulator was activated and an operator gradually adjusted the stimulation amplitude at the APP test interface until the hind legs moved twice per second, which indicated the threshold strength/intensity. After the start-on test, the animals were subjected to muscle training on daily basis. We increased the stimulation amplitude by 3 levels above the threshold intensity, and then the training was shifted to the doctor-training mode, and the stimulation frequency was set at 6 Hz for 30 min. The daily training lasted for 14 days, and electrical stimulation training was stopped. Afterwards, we tested the threshold once a week and the test lasted for 30 days.
Results
Wound recovery after surgery
After the operation, the wound recovery was closely monitored. The animals recovered well after 7 days. There was no fever reaction during the period.
Surgical operability and system stability
During the implantation, the NuStim-specific surgical tool allowed the operator to easily locate a low-threshold stimulation location in the vicinity of the muscle nerve. The surgical tool could determine the implantation location before the NuStim was released. Strictly following surgical steps and using the surgical tools could prevent NuStim from being implanted into the wrong site and could ensure that the implant could be removed and re-implanted whenever necessary. During the stimulation training, the operator could easily adjust the stimulation training protocols by using the operation software of the NuStim system. The whole stimulation system worked well and uneventfully during the experiment and was well-connected, showing that the stimulator had good stability and reliability.
Threshold test
Seven days after implantation, a threshold test was conducted on weekly basis to record the current threshold voltage level. The result showed that the stimulation threshold was stable, indicating that the implant was stable at its implantation site, and the implant location relative to the muscles and nerves did not change significantly. Since the stimulation threshold experienced no significant change, it showed that the adjacent tissues surrounding the implant recovered well after implantation, and the formation of the fiber membrane did not affect the functioning of the product. Threshold test can serve as a long-term monitoring of tissue intactness and product stability. If the threshold increased continuously, it indicated that the product had a rejecting reaction resulting in the fibrous membrane change, which increased the distance between the stimulator and the nerve. In addition, if the product threshold was increasing constantly, it meant the product was unstable at the implantation site. At present, the results on the threshold changes of the product showed that no displacement and rejection took place when the device was properly implanted. The threshold stimulation intensity depended on the location, depth, and neural structure of each animal.
Verification of safety and histocompatibility and stimulation effectiveness
To verify the safety and histocompatibility of the stimulator, it was removed after the experiment ended. The tissues in the vicinity of the stimulator were collected for pathological analysis. We implanted a total of six stimulators into the femoral quadriceps of three dogs, with a non-functional stimulator implanted into a hind leg for comparison.
Light microscopic changes in muscle tissues after implantation
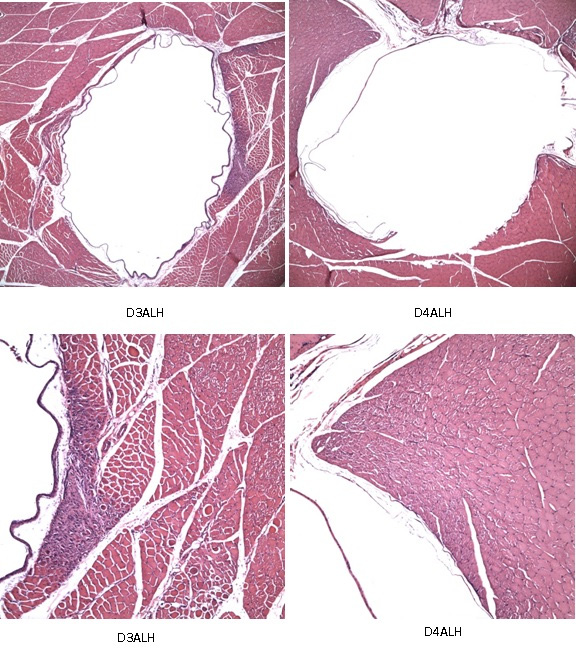
As shown in Figure 3, the implant was surrounded by a thin layer of connective tissue, with a mild cellular reaction. After implantation, the fibrous capsule at one month was found to be approximately twice as thick as that at 3 months. In the vicinity of the fibrous capsule, there was a small amount of muscular tissues with cells containing one central nucleus, indicating the tissues were recovering from the damage caused by initial implantation. The nuclei of distal myofibers were observed distributing around the muscle fibers, presenting a pattern of the healthy muscular fibers. After 3 months, intact muscles were close to the fibrous capsule, indicating that the injured muscles were being constantly repaired. Muscle fibers near the fibrous capsule tended to be transverse (parallel to the long axis of the cylindrical implant). Locally, the implant might have altered the orientation of some muscle fibers.
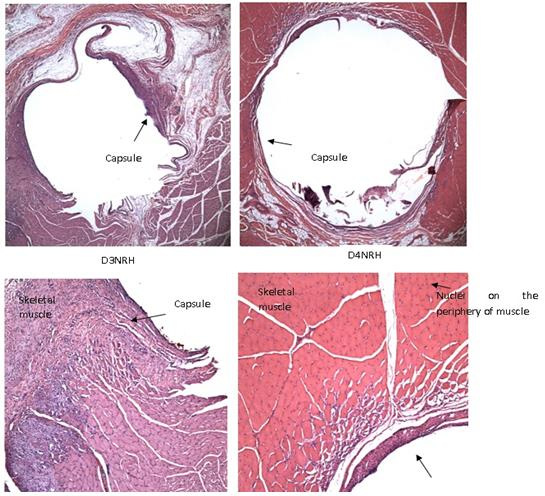
Figure 4 shows that the implant was wrapped by a thin layer of connective tissue with less cellular response. No differences were found between activated and unactivated implants.
Under light microscope, as expected, all stimulators triggered the tissue inflammatory response after the initial implantation. After the inflammatory response disappeared, the connective tissue formed and surrounded the implant to form a capsule, which could stably fix it in the implantation site. Test results of the minimum stimulation intensity that triggered muscle contraction showed that the threshold stimulation intensity was stable, suggesting that once the stimulator was implanted, it did not shift or cause any damage to the nearby tissues. Since the stimulator was made of inert biocompatible materials, no serious tissue damage, such as tissue necrosis, occurred at the implant site.
Figure 3. Section was derived from an activated stimulator: Short-term implanted tissue (1 month) on left and long-term implanted tissue (3 months) on right. The magnifications were 5X and 10X.
Figure 4. Sections was obtained from a unactivated stimulator: Short-term implanted tissue (1 month) on left and long-term implanted tissue (3 months) on right. The magnifications were 5X and 10X.
Figure 5. Normal thigh muscles
Electron microscopic change in the muscle tissue after implantation
As shown in Figure 5, the normal muscle sarcomere was clearly demarcated and the muscle filaments were arranged neatly. No lysis or damage of muscle filaments were seen. Conspicuous Z lines and H lines could be seen. Mitochondria were of circular or oval shape, with high matrix electron density and clear mitochondrial crest (Fig. 5A). The muscle membrane was evenly
smooth, with structurally normal capillaries between muscle cells. The nuclei were normal, and no significant accumulation of glycogen particles was seen (Fig. 5B).
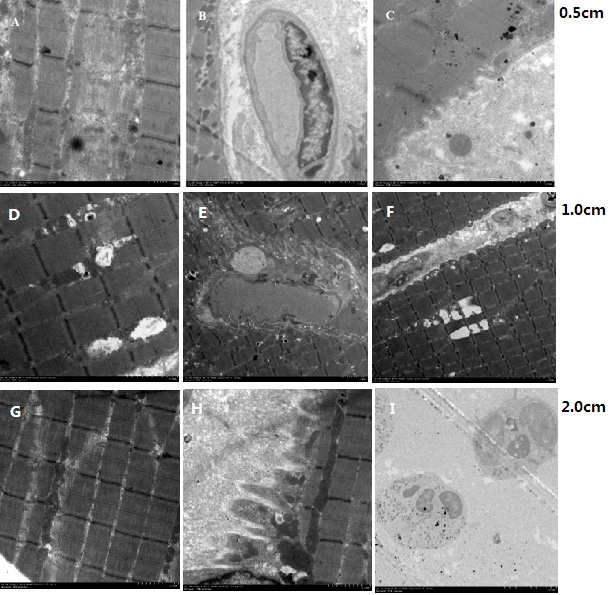
By analyzing the muscle tissue 0.5 cm from the electrode (Fig. 6), we found that the sarcomere boundary was clear, and the distribution of muscle filaments was neat and regular. The local myofilaments were structurally fractured or disappeared. Significant Z lines and H lines could be seen. The mitochondrial crest architecture was unclear, and some mitochondria were structurally destroyed. The number of mitochondria was increased (Fig. 6A). The nuclear structure was normal (Fig. 6B). Some of the myocyte membrane bulged outward, and glycogen aggregation was visible between cells (Fig. 6C).
Muscle tissues 1.0 cm from the electrode (Fig. 6) exhibited clear boundaries, neat distribution of muscle filaments, and mild fracture of local muscle filaments. Z lines and H lines were clear (Fig. 6D). Some of the muscle cell membrane bulged outward, presenting serrated patterns. The nuclear structure was normal (Fig. 6E). Glycogen aggregation was visible between the cells (Fig. 6F).
Figure 6 Section was derived from an stimulation thigh muscle (0.5 cm,1.0 cm,2.0 cm from the electrode)
The muscle tissues 2.0 cm from the electrode (Fig. 6) had clear sarcomere boundaries, neat distribution of myofilaments, and showed no breakage of local myofilaments. Clear Z lines and H lines were noted (Fig. 6G). Some of the myocyte membranes bulged outward and showed a serrated pattern (Fig. 6H). A small number of inflammatory cells were seen between the muscle cells (Fig. 6I).
In short, electron microscopy revealed that electrical stimulation could increase the number of mitochondria and the amount of myoglycogen. It also increased the energy supply and promoted blood circulation. Moreover, the closer a region is to the stimulation electrode, the more the mitochondria and glycogen in it. These findings indicated that the stimulator induced muscle contraction and enhanced muscle motor function. Muscle filament fracture was believed to be caused by muscle damage during sample collection. Moreover, no other obvious abnormal changes were seen, and the stimulator was uneventfully implanted into the body.
Discussion
SUI, as defined by the International Continence Society, is the complaint of involuntary leakage during effort or exertion or during sneezing or coughing [
5]. In questionnaire surveys, most adult women reported that they leak drops of urine with physical exertion from time to time. In fact, half of young nulliparous women reported occasional leakage with coughing, sneezing or exercise [
6]. Lifestyle interventions can decrease stress urinary incontinence in many women [
7]. These include weight loss, decreasing consumption of caffeine, smoking cessation, among others. Traditionally, both α-agonist medications and estrogens have been used to treat stress incontinence. In early uncontrolled case series, women using various estrogen preparations had less incontinence afterward. Moreover, women assigned to receive estrogen and progesterone not only did not have less leakage, but, in fact, were more likely to experience the onset of incontinence or a deterioration of baseline symptoms [
8]. Serotonin and norepinephrine reuptake inhibitors are being investigated as medications for treating stress urinary incontinence, but, up to now, they are not commercially available. The tension-free vaginal tape (TVT), a polypropylene mid-urethral sling, has been swiftly put into clinical use due to its simplicity, lower learning curve relative to laparoscopic procedures and effective marketing. However, women undergoing TVT were more likely to suffer from adverse events, such as bladder perforation, retropubic hematoma, vascular injuries, graft erosion, wound infection, pain, nerve injury, and so on [
9,
10].
In 1948, Arnold Kegel first reported successful use of pelvic floor muscle training (PMFT) as a behavioral therapy for treating SUI symptoms in female patients. The therapy was designed to enhance muscle volume and contraction strength under increased intra-abdominal pressure [
11]. Since then, it has been found to be able to improve UI symptoms in all types of incontinence without side effects [
12]. Although most PMFT programs can be easily administered by healthcare professionals, it may not be cost-effective but is definitively time-consuming [
13].
Hence, it is necessary to develop a device that allows for home-based Kegel exercises. Our preliminary animal studies showed that the NuStim system successfully satisfied the design requirements. The wound recovered well one week after the treatment. We did not observe any side effects over the experimental period. The implantation process was easy, and the performance of the whole stimulation system was stable and reliable. The stimulation threshold was found to be stable, indicating that the implanted device was well-fixed at its implantation site and the distance relative to the muscle nerve did not change significantly. This is a prerequisite for a new implant and it can work stably in the body. On the basis of the results of light microscopy, we re-confirmed that the stimulator did not shift or cause damage to nearby tissues. The whole system showed a good biocompatibility with recipient animals. In addition, electron microscopy showed that the stimulator could cause muscle contraction and enhance muscle motor function, indicating that the stimulation system could simulate PMFT. We conducted another animal study by using the stimulator to stimulate tibial nerve [
14]. The device implanted by minimally invasive procedure was shown to be effective in inhibiting the micturition reflex under physiological conditions. It further proved that it has potential to be used in clinical practice for the treatment of SUI.
However, the present study has some limitations. First, in the current study, we examined the effects of the stimulation system for only one month, while stimulation is delivered chronically in clinical practice. Thus, the long-term stability and safety warrant further studies. Second, we did not establish an animal model of SUI. Hence, more studies are needed to confirm the effects of the stimulation system in SUI animals. Lastly, multicenter clinical trials are required to verify its clinical efficacy.
Conclusion
Animal tests on the NuStim stimulation system showed that the stimulator was safely implanted into the body, with good tissue compatibility, stability and reliability. It also met the requirements of human clinical trials.