INTRODUCTION
Bladder cancer ranks as the fourth most common cancer in men and is the eighth most common malignancy in women in the United States. The incidence of pelvic lymph nodal metastases in patients with stage T1-T4N0M0 bladder cancer undergoing radical cystectomy (RC) is approximately 25%, correlating with the depth of invasion of the primary bladder tumor. Despite lymph node involvement, nearly 31% of these patients were alive at 5 years and 23% were alive at 10 years. Meticulous extended pelvic lymph node dissection (PLND) might contributed to the long-term survival of many of these patients [
1]. In fact, growing evidence supports more extensive and thorough dissection to enhance nodal yield and ultimately improve local control and survival.
RC is a classic surgical procedure for the treatment of high-grade muscle-invasive and recurrent high-grade organ-confined bladder carcinoma. Increasing evidence suggests that extended PLND during RC not only provides information for tumor staging and prognosis but also have clinically significant therapeutic benefits for both node-positive and node-negative patients [
2].
Compared to its open counterparts, laparoscopic and robotic-assisted surgery allows for precise surgical manipulation, with superior visualization and reduced blood loss. In recent years, numerous case series have demonstrated the technical feasibility of laparoscopic radical cystectomy (LRC) and robotic-assisted radical cystectomy (RRC) in patients with bladder cancer, offering potential advantages such as less surgical blood loss, faster recovery, and earlier hospital discharge [
3]. Since LRC and RRC are newly introduced procedures, it is essential to investigate whether extended PLND, adhering to anatomical boundaries delineated by open surgery, can be performed laparoscopically [
4]. Current literature suggests that robotic-assisted PLND had higher lymph node yield than open surgery and laparoscopic PLND, while laparoscopic PLND had the lowest lymph node yield. Consequently, the fewer nodes being removed via the laparoscopic approach raise oncological concerns [
5].
A comprehensive understanding of the anatomic extent of open PLND plus accumulated experience in laparoscopic and robotic-assisted surgery has made it possible to apply laparoscopic and robotic surgical techniques to RC with extended PLND. Experienced surgeons have been endeavoring to overcome technical barriers associated with the extended node dissection. Some reports demonstrated this approach was feasible [
6]. A non-randomized comparative study also reported similar median total lymph node yields, with 15 in the ORC group and 16 in the RRC group [
7]. LRC and RRC with extended PLND may serve as alternative treatments for high-risk or refractory superficial and muscle-invasive bladder cancer.
In this review, we examined the current data on the technique of laparoscopic and robotic-assisted extended PLND, as well as its the clinical outcomes and surgical complications.
MATERIALS AND METHODS
A systematic review was conducted by searching the electronic databases MEDLINE®/PubMed® using the following keywords: bladder cancer, radical cystectomy, laparoscopy, robotics, and pelvic lymph node dissection. A total of 205 references were retrieved on the two techniques, and 46 articles were selected on the basis of their abstracts, contents, and study designs. The candidate articles were evaluated in terms of their contribution to the field in the following four respects: (1) evolution of pelvic lymph node dissection; (2) development of LRC and RRC with PLND techniques; (3) limiting factors/restraints and complications; and (4) oncological outcomes.
1. Indications and Contraindications of LRC and RRC [6]
1.1 Indications
1.1.1 Stage T2-T4a muscle-invasive bladder cancer.
1.1.2 High-grade, recurrent, non-muscle-invasive bladder cancer.
1.2 Contraindications
1.2.1 Bulky lymphadenopathy.
1.2.2 Locally advanced disease (T4).
1.2.3 Uncorrected coagulopathy.
1.2.4 Morbid obesity (body mass index [BMI] > 35).
1.3 Relative Contraindications
1.3.1 Prior abdominal surgery.
1.3.2 Radiotherapy.
1.3.3 Neoadjuvant chemotherapy.
1.3.4 Severe cardiorespiratory compromise.
2. Techniques of LRC and RRC with extended PLND
2.1 Position and Trocar
Patients were put under general anesthesia, placed in a steep Trendelenburg position, and carefully secured and padded to prevent neuromuscular injury.
A modified version of the classic transperitoneal five-trocar arrangement is employed, with the optic and right-lateral trocars repositioned more cephalically. The optic trocar is introduced in/along the midline, approximately 4 cm (two finger widths) above the umbilicus. The supraumbilical camera position is crucial for adequate visualization of the upper limit of the PLND. The right-lateral trocar is also placed two finger widths above and medially to its original position for use by the surgeon's dominant hand, thereby facilitating the performance of the high extended PLND up to inferior mesenteric artery (IMA) (Fig. 1‒2).
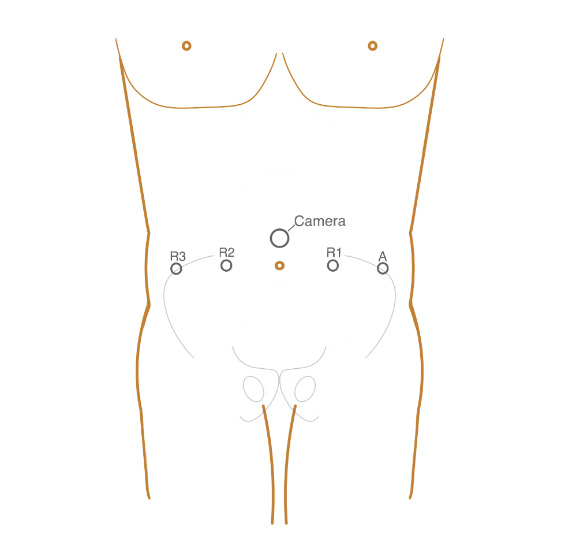
Figure 1. The position of Trocar during robotic-assisted radical cystectomy. A: Trocar for assistant; Camera: Trocar for the camera; R1: Trocar for the first arm of the robot; R2: Trocar for the second arm of the robot; R3: Trocar for the third arm of the robot.
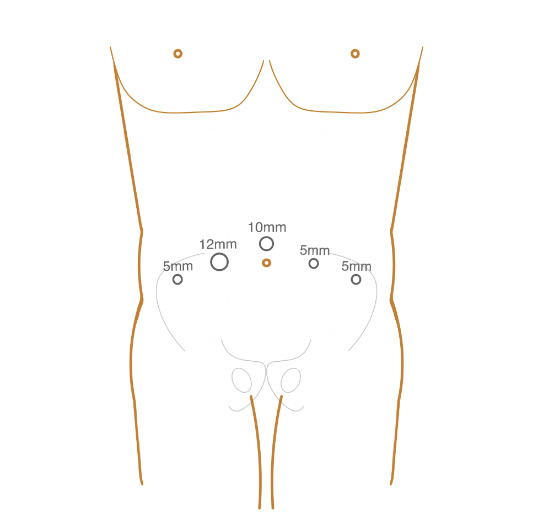
Figure 2. The position of Trocar during laparoscopic radical cystectomy. 12mm: Trocar for ultrasonic scalpel; 10mm: Trocar for the camera; 5mm: Trocar for scissors and forceps.
2.2 Extended PLND Procedure
2.2.1 Initiation with Right Lymphadenectomy
The initial surgical step involves identifying and exposing the external iliac artery and vein. Blunt and sharp dissection of the peritoneum is performed to expose the anterior surface of the external iliac artery. The vein can then be located immediately adjacent (posterior and medial) to the artery. It is essential to dissect into the correct fibroareolar plane just overlying the artery and the vein. Figure 3 shows the extent of extended PLND.
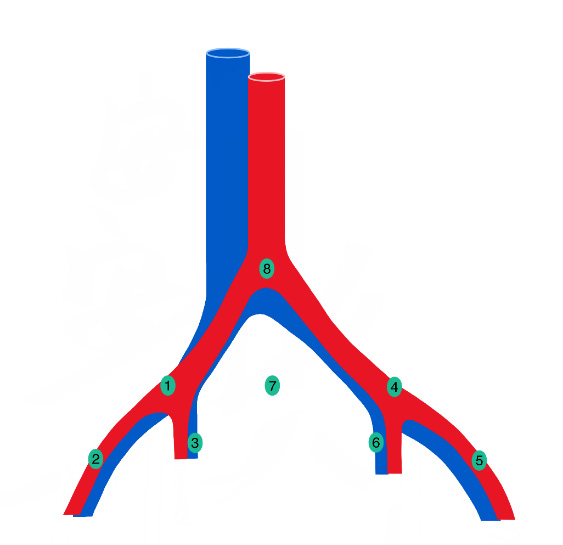
Figure 3. The extent of extended PLND and the location of lymph nodes. 1: Right common iliac lymph node; 2: Right external iliac lymph node; 3: right internal iliac and obturator lymph node; 4: Left external iliac lymph node; 5: Left external iliac lymph node; 6: Left internal iliac and obturator lymph node; 7: Presacral lymph node; 8: Para-aortic lymph node.
2.2.2 Lateral Dissection Border of the Genitofemoral Nerve
Dissection proceeds along the genitofemoral nerve, dividing the fibroareolar tissue and exposing the iliopsoas muscle. The lymphatic tissue packet is entirely lifted en bloc off the surface of the iliopsoas muscle and moved medially.
2.2.3 Anterior Dissection to the External Iliac Artery and Vein
The dissection longitudinally splits and skeletonizes the two vessels circumferentially. It begins along the external iliac vessels from the node of Cloquet up to the aortic bifurcation. Close attention is paid to the location of the collapsed external iliac vein to avoid injury.
2.2.4 Identification and Dissection Along the Obturator Nerve
This step starts by locating and delineating the medial border of the external iliac vein, thereby exposing the obturator fossa posteriorly. With the medial edge of the external iliac vein identified, the plane between the vein and the obturator packet can be extended to the pubic bone distally. Dissection of the umbilical ligament goes down to its origin on the internal iliac artery. The dissection then continues over the internal iliac vessels. Lymphatic tissues from the obturator fossa, including the lymphatics caudal to the node of Cloquet, is clipped and transected. The Triangles of Marcille and tissues along the internal iliac vessels (including the presacral nodes) are removed. Care must be exercised in identifying the circumflex vein distally and any aberrant branches of the external iliac or obturator veins.
2.2.5 Cephalad Dissection Along the Proximal Common Iliac Artery
The circumferentially mobilized common iliac artery is retracted with a vessel loop to completely retrieve fatty tissues in the area distal to the aortic bifurcation. The hypogastric artery is carefully mobilized, with care taken not to injure the internal iliac vein.
2.2.6 Mobilization of the Sigmoid Colon
Optimal exposure necessitates mobilization of the sigmoid colon. This process begins with the dissection of the lateral attachments of the sigmoid colon from the neighboring ureter deep down into the pelvis. The posterior attachments (mesocolon) of the colon are then freed, starting at the sacral promontory and continuing caudally down to the rectosigmoid junction. A similar dissection of the sigmoid colon is performed on the right side.
2.2.7 Dissection and Exposure of Proximal Boundary of the IMA
The boundaries of extended PLND range from the bifurcation of the common iliac vessel to the root of the inferior mesenteric artery. A pelvic peritoneotomy incision is made along the right iliac vessel to the aorta. Dissection proceeds from the bifurcation of the aorta along the surface of the aorta and its left side, cephalad to the root of the inferior mesenteric artery. The psoas muscle is exposed, but care is taken to avoid damaging the lumbar vein. The inferior vena cava is exposed on the right side of the aorta. Dissection continues along the left common iliac artery to the segment below the sigmoid mesentery. The presacral nodes are also removed. Dissection should be gentle to prevent bleeding due to the abundance of venous plexus in the presacral area.
RESULTS
1.Operative outcomes
Table 1 PLND results from laparoscopic radical cystectomy
|
|
|
|
|
|
|
|
| Finelli et al. [8]. 2004 |
Extended |
11 |
N/A |
N/A |
1.5 h LND |
21 (6–30) |
1 deep pelvic vein injury 2 unilateral deep venous thrombosis 1 concomitant pulmonary embolus |
| Haber et al. [9] 2007 |
Limited Extended |
11 26 |
66 (37–81) |
26 (17.4–34.6) |
N/A LND 8.3 (5.5–12) h Total |
6 (2–15) 21 (11–24) |
None |
| Hemal et l. [10] 2008 |
Standard Extended |
48 |
59 (24–80) |
25 (21–33) |
N/A LND 178 (140- 210)Total |
14 (4–24) |
1 external iliac vein injury 1 Deep vein thrombosis 1 Obturator nerve paresis |
| Ghazi et al. [11] 2010 |
Extended |
10 |
63.5 (54–75) |
25.3(21.8–29.1) |
143 (115–165) LND |
25.5 (19–32) |
3 pelvic lympoceles 1 protracted bowel ileus |
| Shao et al. [12] 2010 |
Extended |
43 |
58 (47–71) |
23.6 (19.2–29.1) |
125 (90–185) LND 329(280–470) Total |
31.3 (19–53) |
2 major bleeding 17 lymphatic leakage |
| Huang et al. [13] 2010 |
Standard Extended |
142 29 |
63 (27–84) |
21.3 (15.0–26.4) |
N/A LND 325 (210–605) Total |
13(5–32) 25 (18–46) |
1 Right external iliac vein injury 1 Deep vein thrombosis 11 Lymphatic leakage |
2. Complications
The typical complications associated with LRC and RRC extended PLND include vascular injury, lymphoceles, lymphedema, venous thrombosis, and pulmonary embolism. However, with extended PLND, no increase in morbidity or complication rates was observed .
2.1 The Most Common Intraoperative Complication: Vascular Injury
Clear visualization of anatomic structures during dissection is essential to minimizing the possibility of vessel injuries, especially in the dissection of the para-aortic and paracaval regions. Anatomic landmarks, such as the root of the inferior mesenteric artery and the psoas muscle, can facilitate estimation/identification of the position during dissection. Most injuries to large vessels can be managed laparoscopically by suturing with 5
-0 polypropylene (monofilament), even in the case of the vena cava and iliac artery, under clear vision and with suction and laparoscopic pressure from gauze [
12]. In one case, a left deep pelvic vein injury was controlled with intracorporeal suturing, resulting in a blood loss of 200 mL [
8]. Occasionally, immediate postoperative vascular complications require transfusion and re-operation to control bleeding from a small arterial source, such as the 5-mm pelvic sidewall artery [
14].
2.2 The Second Most Common Postoperative Complication: Lymphatic Leak
Lymphatic leaks often arise from lymphatic vessels that were transected with electrocautery instead of clips during dissection. Postoperatively, clear pelvic lymphatic drainage is frequently observed, and a creatinine level test can exclude a urine leak. Lymphatic leakage typically heals after 2‒3 weeks of drainage without further intervention [
12]. In the cases of persistent leakage, the drainage tube can be removed, allowing the peritoneum to absorb the lymphatic fluid. Even when pelvic lymphoceles occur, they can often be managed conservatively [
11].
2.3 Other Postoperative Complications
Unilateral deep venous thrombosis, including concomitant pulmonary embolism, may develop. Bowel complications are not uncommon and may be associated with persisting pneumoperitoneum and retraction.
DISCUSSION
Radical cystectomy with bilateral PLDN is the standard treatment for high-grade, muscle-invasive bladder cancer, and can achieve optimal recurrence-free and cancer-specific survival [
21]. However, the extent or absolute limits of lymph node dissection have not been well-defined or standardized and remain a subject of debate. Evidence from the literature suggests that a more extended PLND may provide important staging information, therapeutic benefits, and a survival advantage for both node-positive and node-negative patients [
22].
Table 2 PLND results from robotic-asisted radical cystectomy
|
|
|
|
|
|
|
|
| Guru et al. [14] 2008 |
Extended |
58 |
67 (36–90) |
27 (17–45) |
44(19–85) LND |
18 (6–43) |
1 Artery vascular injury |
| Woods et al. [15] 2008 |
Extended |
27 |
67.1 (49–80) |
N/A |
N/A LND 400 (225–660) Total. |
12.3 (7–20) |
3 Deep venous thrombosis |
| Pruthi et al. [16] 2009 |
Standard Extended |
28 22 |
N/A N/A |
N/A N/A |
N/A N/A |
19 (8–33) 30 (12–39) |
None |
| Pruthi et al. [17] 2010 |
Standard |
100 |
65.5 (33–86) |
27.3 |
N/A |
19 (8–40) |
N/A |
| Kasraeian et al. [18] 2010 |
Extended |
9 |
63 (44–82) |
30 (24–32) |
60 LND 270 (210–330) Total |
11 (4–21) |
1 Pelvic hematoma |
| Lavery et al. [19] 2011 |
Extended |
15 |
66 (46–87) |
29 (22–43) |
107 (66–160) LND 423(300–506)Total |
41.8 (18–67) |
None |
| Akbulut et al. [20] 2011 |
Extended |
12 |
60 (43–80) |
24.5 (19.3–31.2) |
N/A LND 10 (8.1–11.5) h Total |
21.3 (8–38) |
1 Right external iliac vein injury |
| Richards et al. [7] 2010 |
Extended |
35 |
65 (59–73) |
27 (23–31) |
N/A LND 530 (458–593) Total |
16 (11–24) |
1 Lymphocele |
1. Anatomic PLND Background
One of the earliest studies reported a long-term survival in approximately 30% of node-positive patients who underwent meticulous LND at RC [
23]. Some researchers labeled removed lymph nodes in terms of their anatomic location of dissection and subsequently analyzed. Thanks to these mapping studies, common sites of pelvic lymph node metastases were identified. Early mapping studies revealed that the lymph node involvement was primarily in obturator, external iliac, common iliac, hypogastric and perivesical nodes, respectively. The detection rate of positive lymph nodes gradually decreased from distal (pelvic) to more proximal (aortic) sites. Nonetheless, the importance of an extended PLND was mentioned by a stage-specific lymph node metastasis mapping study, which included a total of 176 patients who underwent extended PLND during RC. Metastases were found in the lymph nodes of 43 patients (24.4%). Positive lymph nodes were discovered in the perivesical fat and in the pelvic region in 22.7% of the patients, in the common iliac nodes in 8%, in the presacral region in 5.1%, and at or above the aortic bifurcation in 4%. Of patients with pT3 or pT4
, 16% had lymph node metastases outside the common extent for standard PLND [
24]. Most importantly, extended pelvic lymph node dissection could be curative in patients with metastasis or micrometastasis to a few nodes [
25].
1.1 Limited PLND Template
This template encompasses the external iliac vessels and obturator fossa.
1.2 Standard PLND Template
Boundaries for this template include the common iliac artery bifurcation (proximally), the genitofemoral nerve (laterally), the circumflex iliac vein and lymph node of Cloquet (distally), and the hypogastric vessels (posteriorly). The template covers the obturator/hypogastric fossa and the presacral lymph node bilaterally [
26].
1.3 Extended PLND Template
The extended template includes all lymph nodes within the boundaries of the IMA origin from the aorta, the aortic bifurcation and common iliac vessels (proximally), the genitofemoral nerves (laterally), the circumflex iliac vein and lymph node of Cloquet (distally), and the internal iliac vessels (posteriorly). This template comprises the obturator fossa, presciatic (fossa Marcille) lymph nodes, and the presacral lymph nodes overlying the sacral promontory [
27].
1.4 Controversy over Anatomic Extended PLND
Despite substantial data supporting the prognostic and therapeutic value of PLND, no definitive consensus has been reached regarding the cephalad extent of the LND template, even in open surgery. There exists a unanimous agreement that any RC should include an adequate PLND. While the medial, lateral, and distal borders of PLDN have remained consistent, the anatomic extent of PLND is still a subject of debate. The proximal extent has varied from the bifurcation of the common iliac artery to the distal aorta.
Early mapping studies demonstrated that the lymph nodes involved mainly occurred in in obturator, external iliac, common iliac, hypogastric, and perivesical nodes, respectively. In the absence of grossly positive nodes, nodes above the common iliac bifurcation were rarely involved, suggesting that these nodes may not entail a routine resection during RC. Interestingly, up to 16% of lymph node metastases included nodes above the aortic bifurcation, while 8% involved the presacral region. Metastasis to common iliac and more proximal nodes in the absence of pelvic nodal metastasis is uncommon but can occur via the posterior lymphatic collecting ducts, which may drain directly to the common iliac nodes. Proteins associated exclusively with epithelial tissue, such as cytokeratin (CK-19, CK-20) and uroplakin II, have been observed in reportedly negative nodal specimens, indicating that routine microscopic analysis of nodal tissues may miss small foci of metastatic cancer [
28].
Multi-modality single-photon emission computed tomography (SPECT) in combination with computed tomography (CT) and intraoperative gamma probe revealed that the template of the bladder's primary lymphatic landing sites is larger than previously thought. Standard PLND, limited/confined to the ventral portion of the external iliac vessels and obturator fossa, removes only about 50% of all primary lymphatic landing sites. In contrast, extended PLND along the major pelvic vessels, including the internal iliac, external iliac, obturator, and common iliac regions up to the uretero-iliac crossing, removes approximately 90% [
29].
A study compared two consecutive series of 336 patients receiving limited PLND and 322 undergoing extended PLND at RC. The overall lymph node positive rate was 13% in patients with limited PLND and 26% in those who had extended PLND. The 5-year recurrence-free survival in patients with lymph node positive disease was 7% with limited PLND and 35% with extended PLND. Evidence suggests that limited PLND is associated with suboptimal stages and poorer outcomes. Extended PLND allows for more accurate staging and improved survival of patients with non-organ-confined and lymph node positive disease [
30]. Clinical studies showed that primary lymph node metastases went beyond the limit of a standard LND. A group of 290 patients undergoing RC and extended PLDN were prospectively analyzed. The mean total number and standard deviation of lymph nodes removed was 43.1±16.1. Nodal metastases were present in 27.9% of patients. The percentage of metastases at different sites ranged from 14.1% (right obturator nodes) to 2.9% (right paracaval nodes above the aortic bifurcation). In the cases of limited PLND (obturator spaces only), 74% of positive lymph nodes would have been left behind, and roughly 7% of patients would have been under-staged as node-negative. Therefore, it is strongly recommended to perform extended PLDN for all patients to fully remove all metastatic tumor deposits [
31]. Another group/cohort of 336 consecutive patients who underwent RC was prospectively evaluated for lymph node metastasis above the bifurcation of the common iliac vessels. Overall, 34% of patients with lymph node metastases had nodal involvement in the common iliac, periaortic, and presacral regions outside the template of the standard lymph node dissection. When a standard template below the bifurcation of the common iliac vessels were performed, 34% of the lymph node metastases would have been left behind and 6.25% of cases would be incorrectly staged for negative lymphatic metastasis. This finding underscores the argument that extended dissection based on anatomical boundaries, including the common iliac and presacral nodes, not only provides the most accurate staging but also gives the patient the best chance of survival [
32].
2.Development of Laparoscopic Surgery with Extended PLND
Recently, interest has been growing in laparoscopic approaches to extended PLND during RC. The extended PLND template dissection up to the highest proximal extent (IMA) can be consistently accomplished by using both robotic and laparoscopic techniques. Finelli
et al. were the first to report that laparoscopic extended PLND could attain adequate lymph node retrieval. The median number of nodes retrieved was 21 in a group of 11 consecutive patients who underwent extended PLDN. Laparoscopic extended PLDN for bladder cancer can be performed with anatomical boundaries and nodal yields comparable to those of current recommendations for open surgery [
8].
In recent years, robot-assisted approaches have been gaining popularity as a minimally invasive alternative to pure laparoscopic surgery. Robot-assisted laparoscopic surgery enables the surgeon to manipulate the tips of the laparoscopic instruments with a precision similar to that with open surgery, thus facilitating the shifting of standard open surgical procedures into a minimally invasive approach, particularly in the narrow confines of the pelvis. Robot-assisted laparoscopic radical cystectomy (RRC) has emerged as a minimally invasive alternative to pure LRC. Robot-assisted extended PLND during RC can be safely and effectively performed on the robotic platform, yielding results comparable to open cystectomy series at other centers [
19]. The latest findings demonstrated that RRC consistently achieved adequate clearance. The median nodal yield with the extended template up to the IMA was 42.5 (range: 16‒78), which compares favorably with other large series of open RC and satisfies the recommended minimum of 25 lymph nodes needed for an adequate dissection [
33]. Thus, it appears that robot-assisted PLND, utilizing an extended PLND template up to the aortic bifurcation, is technically feasible with comparable nodal yields and intraoperative morbidity similar to open cystectomy series.
3.Restraints of Laparoscopic Surgery with Extended PLND
Although accumulating evidence supports the removal of more lymph nodes via extended LND, as an essential component of the surgical management of patients with muscle-invasive bladder cancer, controversy remains regarding the exact extent of PLND. There is a uniform agreement that when performing LRC or RRC aimed for a cure, it must include adequate laparoscopic PLND that mirrors/ in line with the anatomical boundaries established in open surgery. Despite the increasing use of laparoscopic and robotic RC for the treatment of invasive bladder cancer, valid concerns remain about the feasibility of minimally invasive techniques for achieving adequate nodal clearance, particularly in relation to the associated risks of extended PLND during RC.
3.1 Technical Challenges of LRC
Technically, PLND rarely poses a challenge for laparoscopic or robotic surgeons familiar with the instrumentation, surgical maneuvers, and pelvic anatomy. However, the lack of proprioception and poor ergonomics in a narrow pelvis may compromise crucial steps of the operation. The laparoscopic or robotic technique for PLND should learn open techniques to achieve equivalent oncological results without raising morbidity.
RRC is a similarly complex and demanding procedure that may benefit from the enhanced optical and ergonomic features of the robotic surgical system. Generally, both LRC and RRC resemble the standard ORC procedure. It is a prolonged procedure that includes technical training steps and requires highly developed laparoscopic skills, such as intracorporeal suturing.
Despite the great enthusiasm for new technologies like LRC and RRC, these techniques should be adopted only if they bring clear advantages over existing techniques without compromising traditional surgical standards. Given that a significant percentage of patients have nodal involvement at the time of surgery, patient survival may depend on meticulous PLND during RC. To date, it remains to be determined whether laparoscopic surgery can provide the same potential therapeutic benefits as open surgery by sticking to its principles and anatomical boundaries, and answer to this question requires further investigations in a substantial number of patients on long-term basis.
3.2 Prolonged Operation Time
A significant body of evidence suggests that laparoscopic PLND is technically demanding and requires a longer operation time compared to its open counterpart. Laparoscopic extended PLND has been shown to increase operative time by 1‒1.5 hours when compared to standard PLND at RC [
14].
3.3 Difficulty of Good Exposure
The technical challenge of laparoscopic PLND is most conspicuous in the area of the common iliac vessels, the aortic bifurcation, and up to the IMA, where achieving a good exposure is difficult. Several factors contribute to this problem: the rectosigmoid colon and, to some extent, the small bowel hinder/obstruct the access to the lower aorta, aortic bifurcation, and common iliac vessels. Furthermore, optical or laparoscopic instruments have difficulty accessing tissues above the mid common iliac arteries. These obstacles render simple surgical tasks at the area of the cranial nodes virtually impossible. However, the robotic system's instruments are longer, and the arms have a broader range of motion, improved visibility and magnification, stereoscopic 3-dimensional vision, and fine movements that allow for access to more lymph node zones [
34].
3.4 Lower Lymph Node Yield
More importantly, most studies to date indicate that fewer nodes are removed with laparoscopic PLND than with an open approach, raising oncological concerns. Applying a similar LND template, other researchers reported a median lymph node yield of 18 nodes (Range 6‒43) with laparoscopic LND [
14], and 31.3 nodes (Range 19‒53) with robotic LND [
8]. The laparoscopic or robotic lymph node yields are lower than those of open series from high-volume centers. These open series had a mean lymph node yields of 43 nodes [
31], a mean of 31 nodes (Range 1‒96) with en bloc submission, or a median of 68 nodes (Range 14‒132) with separate nodal packages [
35]. It seems likely that technical difficulties in removing lymphatic tissues along the hypogastric artery and presacral nodes by laparoscopy may be partially responsible for the lower node yield reported with laparoscopic LND. Recently, a randomized prospective trial comparing open to robotic-assisted laparoscopic radical cystectomy with a primary endpoint of lymph node yield found a mean of 18 nodes removed in the open group and 19 nodes removed in the robotic cohort. The robotic approach demonstrated no statistically significant difference in the number of lymph nodes removed compared to the open approach. The robotic approach also compared favorably with the open one in terms of several perioperative parameters, including blood loss and inpatient narcotic requirements [
36].
To investigate the correlation of nodal yield with surgeon experience and surgical volume, the International Robotic Cystectomy Consortium conducted a retrospective multi-institutional study. Of the 527 patients, 437 (82.9%) underwent PLDN. A mean of 17.8 (Range 0‒68) lymph nodes were examined. Surgeon volume was most significantly associated with PLDN on multivariate analysis. High-volume surgeons (> 20 cases) were practically three times more likely to perform PLDN than lower-volume surgeons. The decision to perform a PLDN was ultimately dependent on the responsible surgeon and was mandated by familiarity with anatomy and skill in perivascular dissection [
37].
3.5 The comparison between laparoscopic and robotic extended PLND
Generally, differences exist between LRC and RARC due to factors such as the learning curve, surgeon's experience, and surgical equipment performance. Recent research demonstrated that the primary distinction between laparoscopic extended PLND and robotic extended PLND lies in lymph node yield. Robotic extended PLND has a higher lymph node yield than laparoscopic extended PLND. Consequently, the robot-assisted method led to more accurate staging after RC and PLND [
38]. This is likely attributable to the higher probability of adequate dissection in robotic extended PLND than in laparoscopic extended PLND. The robotic system is equipped with stable mechanical arms and leaves no dead space within the cavity. In confined spaces, the robotic system's high-definition three-dimensional visual system more clearly visualizes anatomical structures, thereby outperforming laparoscopic surgery.
4. Oncological Outcomes of Laparoscopic Surgery with Extended PLND
4.1 Factors Associated with Oncological Outcomes
Removing large numbers of lymph nodes lowers the likelihood of leaving/missing positive nodes undetected, resulting in more favorable prognoses for patients undergoing extended PLND, compared to those receiving limited PLND, in whom some positive nodes might go undetected. Recent studies suggested that the anatomical/dissection extent of PLDN and the number and density of lymph nodes in metastatic disease are crucial independent prognostic factors for bladder cancer.
4.1.1 Number of Lymph Nodes
In patients with nodal metastasis, the number of nodes removed, the number of positive nodes, and the percentage of positive nodes may all be independently predictive of recurrence and survival. Expanding the boundaries of lymph node dissection increases nodal yield, improves staging accuracy, and has been shown to enhance survival. Some researchers recommend excising a predetermined minimum number of lymph nodes (10‒14) as a surrogate quality measure of lymphadenectomy or for pathological examination for staging purposes [
39]. Bladder cancer patients who had positive common iliac lymph node metastasis removed during RC demonstrated similar outcomes to those with nodal disease confined to the true pelvis. The number of positive lymph nodes was an independent predictor of worse oncological outcomes [
40]. In a prospective comparison of robotic versus open RC series, equivalent numbers of lymph nodes were removed in the robotic and open cohorts (17 and 20), and the result indicated that the robotic approach allowed for an upper common iliac node dissection and that the total number of lymph nodes removed was not reduced compared to the open technique [
41].
Unfortunately, translating the findings of different LND templates into a node count that can serve as a criterion for comparing different LND patient series is problematic. Numerous potentially confounding factors, including the surgeon's skills, specimen presentation (packets versus
en bloc) to the pathologist, specimen processing, and methodology of counting nodes, all reportedly impacted the number of lymph nodes removed and enumeration of node metastases [
35].
4.1.2 Lymph Node Density
Lymph node density is defined as the number of lymph nodes involved with the tumor divided by the total number of lymph nodes removed. Patients with a lymph node density of 20% or less enjoyed a 10-year recurrence-free survival rate of 43% , against a mere 17% survival rate at 10 years when lymph node density exceeded 20%. Hence, lymph node density may work better in stratifying lymph node-positive cases since it accommodates both the total number of positive lymph nodes (tumor burden) and the total number of lymph nodes removed (extent of lymphadenectomy) [
42].
A retrospective review of the medical records of 248 patients with lymph node metastases at the time of RC plus PLND for bladder cancer suggested that LND out-did TNM nodal status in predicting disease-specific survival for patients with lymph node-positive disease after RC, even in the context of adjuvant chemotherapy [
43]. In anothr study, clinical and histopathological data from 477 patients with positive lymph node bladder cancer were analyzed, with results supporting the prognostic relevance of lymph node density (threshold value = 20%) in the prediction of cancer-specific survival for both limited PLND and standard/extensive PLND [
44]. A multi-institutional retrospective study showed that lymph node density was a significant prognostic marker. While the number of lymph nodes removed and the raw number of positive lymph nodes may depend on the quality and extent of lymph node dissection, lymph node density may be less influenced by surgical quality [
45]. Despite compelling evidence, similar to total lymph node count, lymph node density relies heavily on the surgeon's ability or preferences, patient's disease burden, and non-standardized pathological evaluation.
4.2 Oncological Outcomes of Laparoscopic Surgery
The feasibility and efficacy of extended PLND in LRC and RRC are well-established. More focus should be directed on extended PLND to consistently achieve higher node yields. From an oncological point of view, intermediate oncological outcomes of LRC and RRC are promising/encouraging and comparable to contemporary open RC series [
46].
The first laparoscopic oncological outcomes were available from 37 patients undergoing LRC followed up for ≤5 years. Overall, the median (range) number of lymph nodes excised was 14 (2‒24). An extended PLND was used in 26 patients (70%), with an increased median yield of 21 (11‒24) nodes. The 5-year actuarial overall, cancer-specific, and recurrence-free survival rates were 63%, 92%, and 92%, respectively. The data suggested that LRC with extended PLND, following established oncological principles could achieve oncological outcomes comparable to those attained in contemporary series of open RC [
9].
Recent studies reported laparoscopic recurrence-free survival rates ranging from 83% to 85% at 1 to 2 years and 60% to 77% at 2 to 3 years, while robotic-assisted studies reported recurrence-free survival rates of 86% to 91% at 1 to 2 years [
47]. The early survival rates among unselected bladder cancer patients treated with RRC were as follows: extended PLDN was performed in 98% of patients, and disease-free, cancer-specific, and overall survival rates at 2 years were 74%, 85%, and 79%, respectively. Early survival outcomes were similar to those reported in recent open RC series, with a notably low incidence of local recurrence [
48]. The oncological outcomes of LRC for 171 cases with a median follow-up of 37 months (3‒83) revealed that 54 patients (31.6%) completed a 5-year follow-up. The estimated 5-year overall survival, cancer-specific survival, and recurrence-free survival rates were 73.7%, 81.3% and 72.6%, respectively [
13]. Although the adoption of minimally invasive approaches for RC is on the rise, the long-term functional and oncological outcomes of these techniques have yet to be proven. Prospective controlled studies and long-term follow-up are needed to determine the proper use of laparoscopic and robotic-assisted techniques in RC with extended PLDN.
4.3 Patients Eligible for Extended PLND
The available evidence indicates that radical cystectomy with an extended PLND can enhance overall survival and cancer-specific survival [
49]. Pathological findings from extended PLND specimens provide valuable prognostic data that inform treatment decisions for this patient population. However, as a more extensive lymph node dissection, the technique may lead to lengthier surgical time and higher complication rates, and high-risk patients typically undergo standard or limited lymph node dissection. Conversely, patients with fewer comorbidities and good functional status are suitable candidates for an extended PLND [
50]. Additionally, there is evidence to suggest a therapeutic benefit of extended PLND in patients with ≥ pT3 disease, regardless of lymph node status (positive or negative) [
51].
CONCLUSIONS
A growing body of evidence suggests that LRC and RRC with extended PLND are feasible and safe, with low perioperative morbidity but longer operative time compared to open surgery. The short-term oncological outcomes appeared to be comparable to those of the open approach. Despite increasing interest in minimally invasive techniques, they should be considered investigative at this stage, and a systematic learning approach to these procedures will serve one well in accomplishing a successful, anatomic PLND, reducing the risk of morbidity and complications. In the future, long-term survival studies are needed to confirm the oncologic efficacy of LRC and RRC with extended PLND for the treatment of invasive bladder cancer.





